Microarrays (CytoSure)
Microarrays for cytogenetics, rare diseases and cancer research
- 20+ years experience in the development of microarrays
- Excellent CNV and LOH calling, down to exon level
- CytoSure Constitutional v3: the ONLY array with the latest ID/DD gene content (ClinGen/DDD)
- Customised arrays for desired content and format flexibility
- Straightforward and fast data analysis with complimentary CytoSure Interpret Software
Why are Microarrays still relevant today?
It is well known that several inherited diseases are a consequence of genetic rearrangements such as gene duplications, translocations and deletions. These alterations are observed in cancer cells. (1)
The year 2004 marked a milestone for genetics, with the implementation of array comparative genomic hybridisation (aCGH) into clinical research. Since then, high quality arrays, standardised hybridisation protocols, accurate scanning technologies and robust computational methods have established DNA microarray as a powerful, mature and easy-to-use essential genomic tool. (1) Nowadays, DNA microarrays are recognised as first-tier tests for developmental delay/intellectual disability, autism spectrum disorders, multiple congenital anomalies/dysmorphic features, cancers as well as prenatal detection of clinically relevant copy number variations in foetuses. (2)
Developmental delay (DD)/intellectual disability (ID), characterised by a significant impairment of cognitive and adaptive functions, affects 1–3 % of the population with most affected individuals remaining without proper diagnosis. The aetiology of DD/ID is heterogeneous with both genetic and environmental contribution. High-resolution G-banded karyotype reveals chromosome abnormalities in 3–5 % of patients and molecular cytogenetics, e.g. fluorescence in situ hybridisation (FISH) provides diagnosis in an additional 3–6 % of cases. In the past few years, application of aCGH and single-nucleotide polymorphism (SNP) arrays has revolutionised the clinical research of DD/ID. (3)
Ready to move your ID/DD research to the next level? Check our CytoSure® Constitutional NGS solution (pdf).
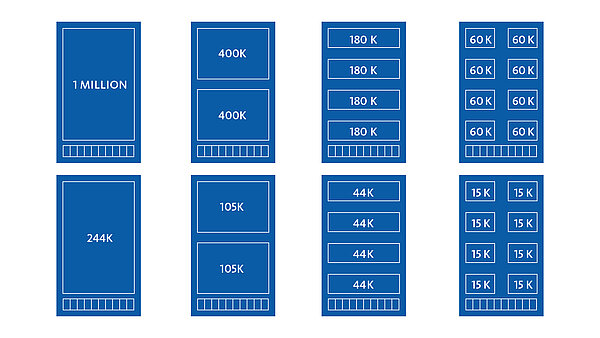
The CytoSure® product portfolio includes DNA microarrays, genomic DNA labelling kits, next-generation sequencing tools and analysis software.
What is the CytoSure offering?
CytoSure® is a range of products that have been developed with input from leaders in the fields of cytogenetics, cancer and rare disease research – which have been optimised to provide the most relevant content and sensitivity.
Our arrays are supported by complimentary analytical software – CytoSure Interpret – our proprietary software package that has been designed to be adaptable and user-friendly.
How can CytoSure help you overcome daily challenges?
- CytoSure® Constitutional v3 arrays combine accurate identification of CNV, LOH, UPD and consanguineous samples, all on a single array for cost-effective detection of a broader range of genetic syndromes
- High-resolution exon coverage of targeted genes for autism, epilepsy, cardiomyopathy, skeletal dysplasia, metabolic disorder and other rare diseases so that the mutation spectrum of these phenotypically and genetically heterogenous diseases are efficiently interrogated
- Versatile cancer arrays with unique SNP probe technology so that any reference sample without restriction digest can be used
- Custom designed arrays so that your unique requirements on content and format are fulfilled with maximum flexibility
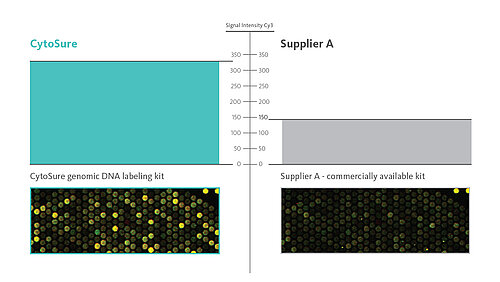
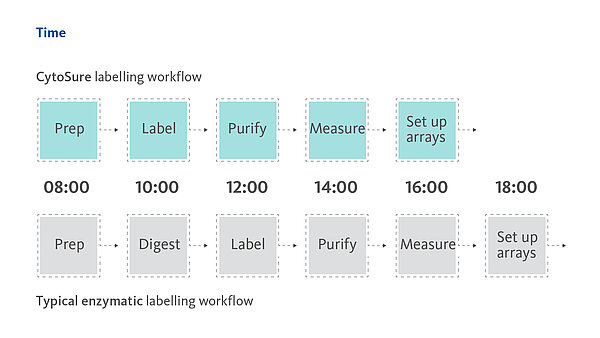
- Optimised genomic DNA labelling kits rapidly delivering high signal-to-noise ratios so that you can be confident on your data accuracy and reporting
- Industry-leading powerful and easy-to-use package – CytoSure® Interpret Software – so that your oligo aCGH data translates into meaningful results, effortlessly
- Continuous expert consultation by our medical affairs and application team so that you can be reassured to achieve the best outcome from your array, every time
References
[1] Trevino, V., Falciani, F. & Barrera-Saldaña, HA. DNA Microarrays: A Powerful Genomic Tool for Biomedical and Clinical Research. Mol Med 13, 527–541 (2007). https://doi.org/10.2119/2006-00107.Trevino
[2] Cheung SW, Bi W. Novel applications of array comparative genomic hybridization in molecular diagnostics. Expert Rev Mol Diagn, 18(6):531-542 (2018). doi: 10.1080/14737159.2018.1479253
[3] Bartnik, M., Nowakowska, B., Derwińska, K., et al. Application of array comparative genomic hybridization in 256 patients with developmental delay or intellectual disability. J Appl Genetics 55, 125–144 (2014). https://doi.org/10.1007/s13353-013-0181-x
CytoSure®: For Research Use Only, not for use in diagnostic procedures. Product availability may vary from country to country and is subject to varying regulatory requirements. Contact your local representatives for availability.
Manufacturer and trademarks: CytoSure® (Oxford Gene Technology IP Limited)
CytoSure Interpret Software requirements:
The recommended IT requirements results in one-hour analysis time for each sample
| Requirement | Minimum | Recommended |
| Memory (RAM) | 16 GB | 24 GB |
| CPU Cores | 8 | 16 |
| Storage (Disk space) | 500 GB | 2 TB |
| Operating System | Windows 7 or newer | Windows 7 or newer |
| Other | Virualisation (VT-x) to be enabled in the BIOS | Virualisation (VT-x) to be enabled in the BIOS |
Sysmex Nordic ApS Suomen sivuliike
KPY Novapolis
Microkatu 1
70210 Kuopio
Finland
+358 (0) 102999940